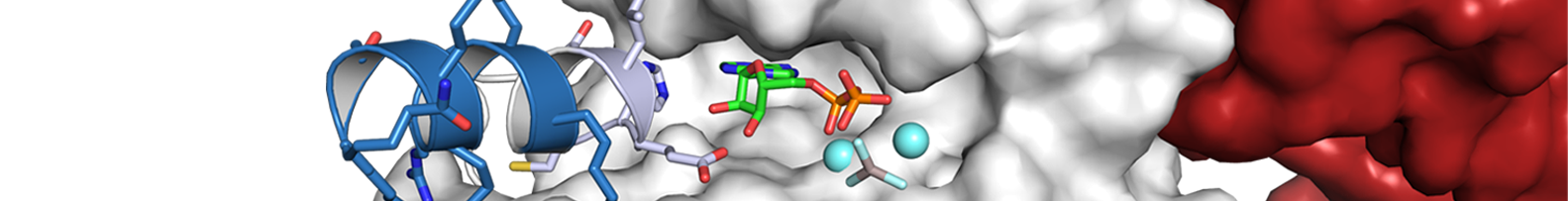
The immune system is permanently defending the human body against a wide range of different pathogens. A common response to these harmful stimuli is inflammation. The cytosolic receptor protein NLRP3 is one member of the family of Nod-like receptors (NLRs). NLRP3 recognizes pathogen-associated and danger-associated molecular patterns in immune cells. These molecular patterns lead to an activation of NLRP3 and the formation of a high-molecular assembly, called inflammasome. NLRP3 is a multi-domain protein of 120 kDa in size that is expressed in myeloid cells as, e.g., macrophages. We are analysing the structure of NLRP3 as well as other NLRs and correlate structural features with the function and activity of these inflammatory receptors. A broad variety of molecular, biochemical, and structural techniques is applied to study the effects of activation and nucleotide binding in NLRs. The correlation of structure–function relationships in NLRP3 will provide insights into the molecular assembly of the inflammasome and yield key sites for the regulation and targeted inhibition of this immune receptor.
The immune system is permanently defending the human body against a wide range of different pathogens. A common response to these harmful stimuli is inflammation. The cytosolic receptor protein NLRP3 is one member of the family of Nod-like receptors (NLRs). NLRP3 recognizes pathogen-associated and danger-associated molecular patterns in immune cells. These molecular patterns lead to an activation of NLRP3 and the formation of a high-molecular assembly, called inflammasome. NLRP3 is a multi-domain protein of 120 kDa in size that is expressed in myeloid cells as, e.g., macrophages. We are analysing the structure of NLRP3 as well as other NLRs and correlate structural features with the function and activity of these inflammatory receptors. A broad variety of molecular, biochemical, and structural techniques is applied to study the effects of activation and nucleotide binding in NLRs. The correlation of structure–function relationships in NLRP3 will provide insights into the molecular assembly of the inflammasome and yield key sites for the regulation and targeted inhibition of this immune receptor.